AED 2.10
Description
Thickness: the copper thickness of 0.3mm
chemistry is the driving force behind the magic of batteries. A battery is a package of one or more galvanic cells used for the production and storage of electric energy by chemical means. A galvanic cell consists of at least two half cells, a reduction cell and an oxidation cell. chemical reactions in the two half cells provide the energy for the galvanic cell operations.
Each half cell consists of an electrode and an electrolyte solution. Usually the solution contains ions derived from the electrode by oxidation or reduction reaction.
A galvanic cell is also called a voltaic cell. The spontaneous reactions in it provide the electric energy or current.
Two half cells can be put together to form an electrolytic cell, which is used for electrolysis. In this case, electric energy is used to force nonspontaneous chemical reactions.
Oxidation Reduction Reactions
Many definitions can be given to oxidation and reduction reactions. In terms of electrochemistry, the following definition is most appropriate, because it let's us see how the electrons perform their roles in the chemistry of batteries.
Oxidation and reduction reactions cannot be carried out separately. They have to appear together in a chemical reaction. Thus oxidation and reduction reactions are often called redox reactions. In terms of redox reactions, a reducing agent and an oxidizing agent form a redox couple as they undergo the reaction:
Oxidant+ne−→Reductant
Reducant→Oxidant+ne−
An oxidant is an oxidizing reagent, and a reductant is a reducing agent. The
reductant | oxidant or oxidant | reductant
Two members of the couple are the same element or compound, but of different oxidation state.
Copper-Zinc Voltaic Cells
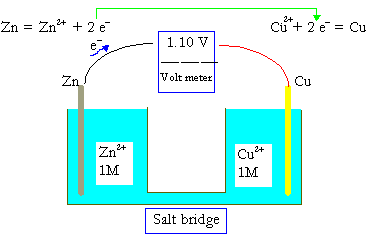 As an introduction to electrochemistry let us take a look at a simple voltaic cell or a galvanic cell.
As an introduction to electrochemistry let us take a look at a simple voltaic cell or a galvanic cell.
When a stick of zinc (Zn) is inserted in a salt solution, there is a tendency for Zn to lose electrons according to the reaction,
Zn→Zn2++2